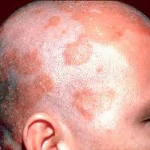
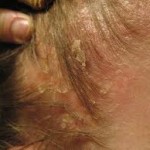
Capex® Shampoo (fluocinolone acetonide) topical shampoo, 0.01% is a unique corticosteroid treatment in a shampoo-delivery system created to effectively treat inflammatory diseases of the scalp, such as itch and dandruff associated with seborrheic dermatitis (SD).When used every day for two weeks, Capex® Shampoo treats inflammatory diseases related to SD. Some side effects have been reported infrequently in people who have used topical steroids, such as inflammation, burning, itching and irritation.
DESCRIPTION:  Capex® Shampoo 0.01% is supplied as a shampoo formulation with a 12 mg fluocinolone acetonide capsule which is emptied into the shampoo base by the pharmacist at the time of dispensing. After mixing, Capex® Shampoo contains fluocinolone acetonide (6a, 9-Difluro-11b, 16a, 17,21  -tetrahydroxypregna-1,4-diene-3, 20-dione cyclic 16,17-acetal with acetone), a synthetic fluorinated corticosteroid for topical dermatologic use. The corticosteroids constitute a class of primarily synthetic steroids used topically as an anti-inflammatory and antipruritic agents.Chemically, Capex® Shampoo mixture is C24H30F2O8.Fluocinolone acetonide in Capex®
Shampoo has the molecular weight of 452.50. It is a white crystalline powder that is odorless, stable in light and melts at 270°F with decomposition; soluble in alcohol, acetone and methanol; slightly soluble in chloroform; insoluble in water.Each fluocinolone capsule contains 12 mg of fluocinolone acetonide, 548 mg of dibasic calcium phosphate dihydrate USP, and 240 mg of talc USP. The shampoo  base contains aluminum acetate basic, benzalkonium chloride solution, boric acid, citric acid anhydrous, cocamido-ether-sulfate complex, cocoamine oxide,  lauramide DEA, magnesium aluminum silicate, methylparaben, oat flour, propylene glycol, propylparaben, purified water, and fragrances, with D&C Yellow #10 and FD&C Blue #1 as coloring.
Why should I use Capex shampoo?
- no tar or sulfur smell
- easy application: once a day for five minutes (for first two weeks)
- low incidence of side effects
- reduction of inflammation, itching and scaling
How do I use Capex shampoo?
- Shake Capex® Shampoo bottle well.
- Wet the hair and scalp thoroughly, apply sufficient amount of Capex® Shampoo, and massage into the scalp. (Please note that this product does not contain drying detergents and therefore produces  minimal lathering.)
- Allow Capex® Shampoo to remain on the scalp for five minutes. (Note: It is important to keep the shampoo on for five minutes for effective treatment.)
- Rinse thoroughly with water. Conditioner is not necessary.
- Repeat these instructions daily for two weeks. Following the initial two weeks, one treatment per week is usually       sufficient to keep the itching and flaking of dandruff from returning. Consult your physician.
CLINICAL PHARMACOLOGY: Like other topical corticosteroids, fluocinolone acetonide has anti-inflammatory, antipruritic and vasoconstrictive properties. The mechanism of the anti-inflammatory activity of the topical steroids, in general, is unclear. However, corticosteroids are thought to act by the induction of phospholipase A2 inhibitory proteins, collectively called lipocortins. It is postulated that these proteins control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes by inhibiting the release of their common precursor arachidonic acid. Arachidonic acid is released from membrane phospholipids by phospholipase A2.
Pharmacokinetics: The extent of percutaneous absorption of topical corticosteroids is determined by many factors including the vehicle and the integrity of the epidermal barrier. Occlusive dressings with hydrocortisone for up to 24 hours have not been demonstrated to increase penetration; however, occlusion of hydrocortisone for 96 hours markedly enhances penetration. Topical corticosteroids can be absorbed from normal intact skin while inflammation and/or other disease processes in the skin increase percutaneous absorption.
Capex® Shampoo is in the low- to medium-potency as compared with other topical corticosteroids.
CLINICAL STUDIES: In vehicle-controlled studies for the treatment of Seborrheic Dermatitis of the scalp, after 14 days of treatment, 84% of patients on active treatment and 29% of patients on the drug vehicle had cleared or markedly improved.
INDICATION AND USAGE: Capex® Shampoo is a low- to medium-potency corticosteroid indicated for the treatment of Seborrheic Dermatitis of the scalp. This product has not been proven to be effective in other corticosteroid-responsive dermatoses.
CONTRAINDICATIONS: Capex® Shampoo is contraindicated in those patients with a history of hypersensitivity to any of the components of the preparation.
PRECAUTIONS: General: Systemic absorption of topical corticosteroids can produce reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticoid insufficiency after withdrawal of treatment. Manifestations of Cushing’s syndrome, hyperglycemia, and glucosuria can also be produced in some patients by systemic absorption of topical corticosteroids while on treatment.
Patients applying a topical steroid to a large surface area or to areas under occlusion should be evaluated periodically for evidence of HPA axis suppression. This may be done by using the ACTH stimulation, A.M. plasma cortisol, and urinary free cortisol tests. Patients receiving superpotent corticosteroids should not be treated for more than 2 weeks at a time and only small areas should be treated at any one time due to the increased risk of HPA suppression.