In addition, there is an accommodating lens that was approved by the US FDA in 2003 and made by Eyeonics, now Bausch & Lomb. The Crystalens (R) is on struts and is implanted in the eye’s lens capsule, and its design allows the lens’ focusing muscles to move it back and forth, giving the patient natural focusing ability.
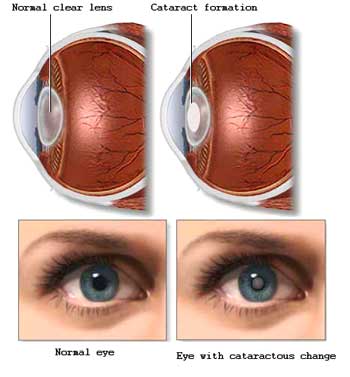
Artificial intraocular lenses are used to replace the eye’s natural lens that is removed during cataract surgery. These lenses have been increasing in popularity since the 1960s, but it was not until 1981 that the first U.S. Food and Drug Administration (FDA) approval for this type of products was issued. The development of intraocular lenses brought an innovation into the optical world as before they could be used; patients would not have their natural lens replaced and as a result, they had to wear very thick eyeglasses or some special type of contact lenses. Nowadays, IOLs are especially designed for patients with different vision problems. The main types of IOLs that now exist are divided into monofocal and multifocal lenses.
The monofocal intraocular lenses are the traditional ones, which may provide vision at one distance only: far, intermediate, or near. Patients who choose these lenses over the more developed types will have to overcome the disadvantage of wearing eyeglasses or contact lenses for reading or using the computer. These intraocular lenses are usually spherical, and they have their surface uniformly curved.
The multifocal intraocular lenses are ones of the newest types of such lenses. They are often referred to as “premium” lenses because they are multifocal and accommodative and allow the patient to visualize objects at more than one distance, removing the need to wear eyeglasses or contact lenses. Premium intraocular lenses are those used in correcting presbyopia or astigmatism. Premium intraocular lenses are typically not covered by insurance companies as their additional benefits are considered a luxury and not a medical necessity. An accommodative intraocular lens implant has only one focal point, but it acts as if it is a multifocal IOL. The intraocular lens was designed with a hinge similar to the mechanics of the eye’s natural lens.
 The intraocular lenses used in correcting astigmatism are called toric and have been FDA approved since 1998. The STAAR Surgical Intraocular Lens was the first such lens ever developed in the United States and it may correct up to 3.5 diopters. A different model of toric lenses is created by Alcon and may correct up to 3 diopters of astigmatism. In order to achieve the most benefit from a toric lens, the surgeon must rotate the lens to be on axis with the patient’s astigmatism. Interoperative wavefront analysis, such as that provided by the ORA System developed by Wavetec Visions Systems can be used to assist the doctor in Toric lens placement and minimize astigmatic errors.
The intraocular lenses used in correcting astigmatism are called toric and have been FDA approved since 1998. The STAAR Surgical Intraocular Lens was the first such lens ever developed in the United States and it may correct up to 3.5 diopters. A different model of toric lenses is created by Alcon and may correct up to 3 diopters of astigmatism. In order to achieve the most benefit from a toric lens, the surgeon must rotate the lens to be on axis with the patient’s astigmatism. Interoperative wavefront analysis, such as that provided by the ORA System developed by Wavetec Visions Systems can be used to assist the doctor in Toric lens placement and minimize astigmatic errors.
Cataract surgery may be performed to correct vision problems in both eyes, and in these cases, patients are usually recommended to consider monovision. This procedure involves inserting in one eye an intraocular lens that provides near vision and in the other eye an IOL that provides distance vision. Although most patients can adjust to having implanted monofocal lenses in both eyes, some cannot and may experience blurred vision at both near and far. IOLs that emphasize distance vision may be mixed with IOLs that emphasize intermediate vision in order to achieve a type of modified monovision. Bausch and Lomb developed in 2004 the first aspheric IOLs which provide better contrast sensitivity by having their periphery flatter than the middle of the lens. However, some cataract surgeons have debated the benefits of aspheric IOLs, because the contrast sensitivity benefit may not last in older patients.
 Some of the newly launched IOLs are able to provide ultraviolet and blue light protection. The crystalline lens of the eye filters these potentially harmful rays and many premium IOLs are designed to undertake this task as well. According to few studies though, these lenses have been associated with a decrease in vision quality.
Some of the newly launched IOLs are able to provide ultraviolet and blue light protection. The crystalline lens of the eye filters these potentially harmful rays and many premium IOLs are designed to undertake this task as well. According to few studies though, these lenses have been associated with a decrease in vision quality.
Another type of intraocular lenses is the light-adjustable one which is still undergoing FDA clinical trials. This particular type of IOLs is implanted in the eye and then treated with light of a certain wavelength in order to alter the curvature of the lens.
In some cases, surgeons may opt for inserting an additional lens over the already implanted one. This type of IOLs procedures are called “piggyback” IOLs and are usually considered an option whenever the lens the result of the first implant is not optimal. In such cases, implanting another IOL over the existent one is considered safer than replacing the initial lens. This approach may also be used in patients who need high degrees of vision correction.